
Operating at the Intersection of Regulation, Science, and Execution
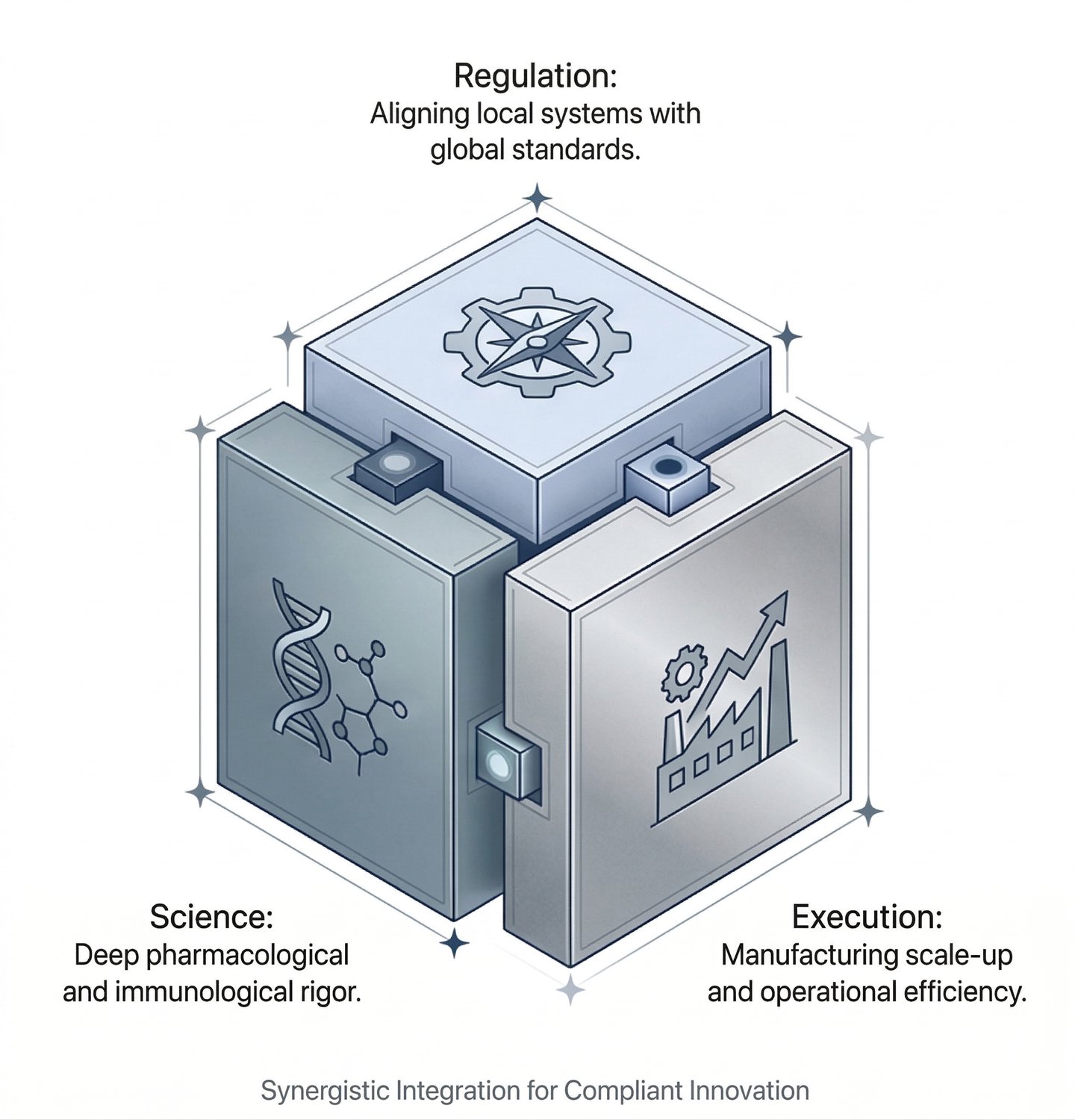
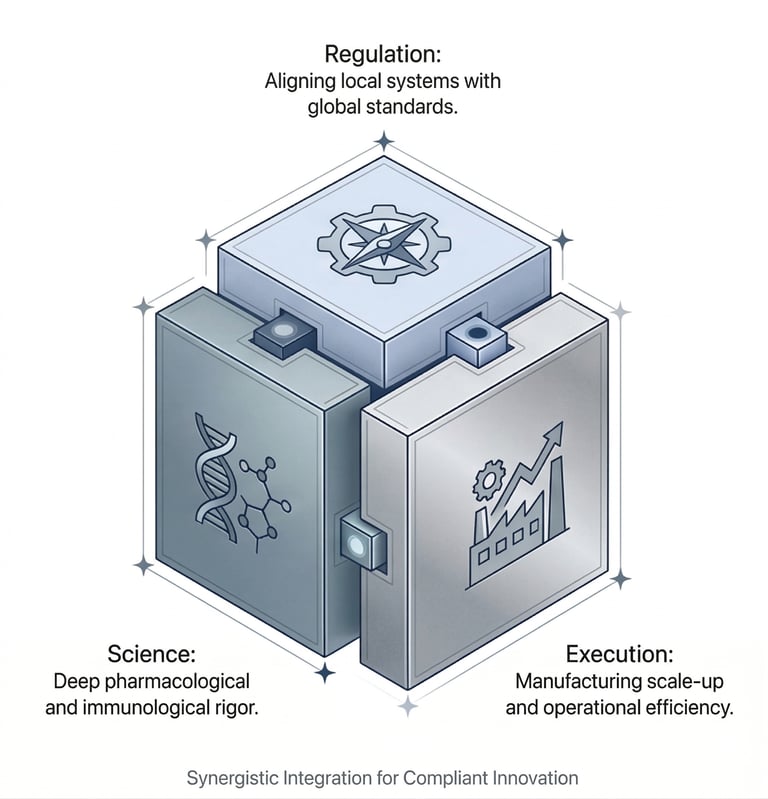
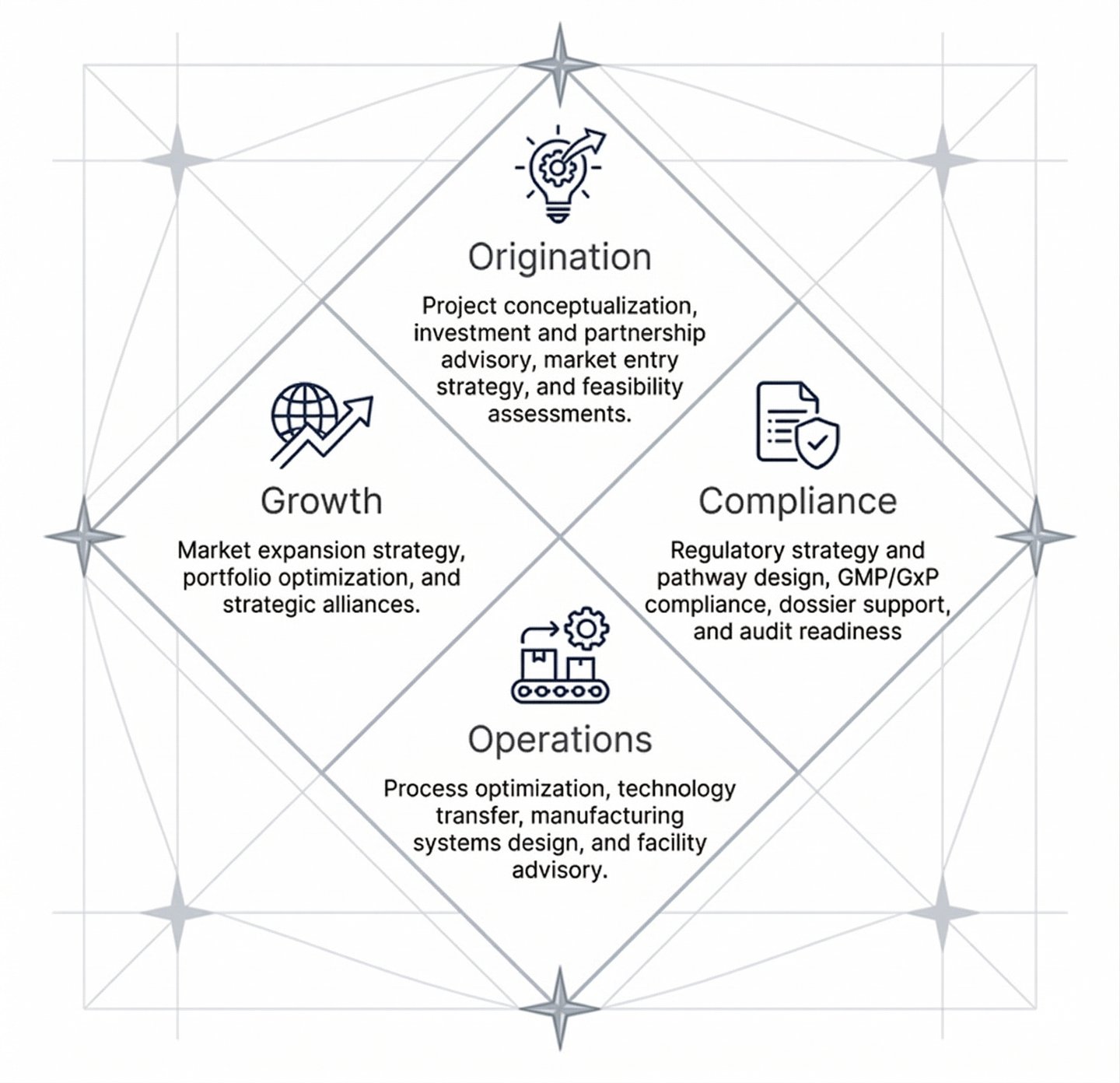
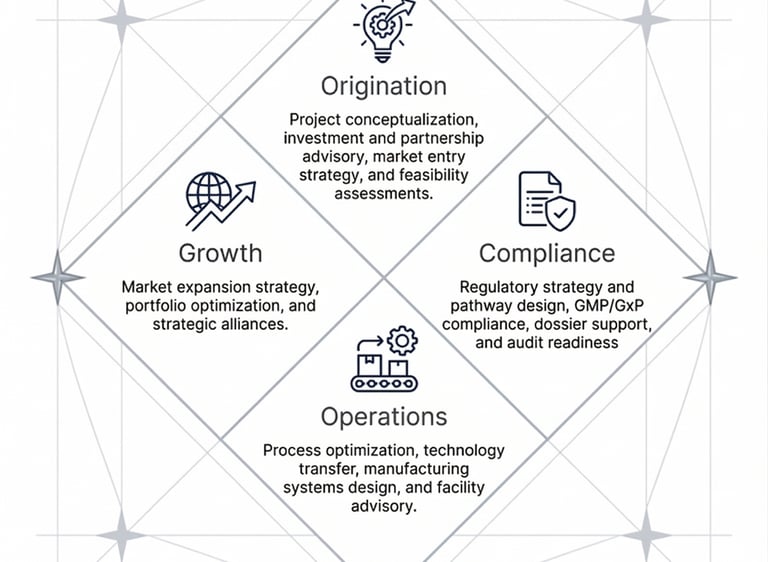
The Four Pillars of Our Service Delivery
Cardinal Pharma structures its value proposition around four critical phases of the pharmaceutical lifecycle
Stay Informed
Get updates on pharma compliance and innovation